 Photo Shutterstock
Photo Shutterstock
Health Canada is recalling several drugs used to treat high blood pressure that help prevent heart attacks and strokes.
The drugs contain valsartan and are being recalled by their manufacturers because the valsartan was found to contain an impurity callled N-nitrosodimethylamine (NDMA).
NDMA is a potential carcinogen and can cause cancer with long-term exposure.
Health Canada says people should keep taking their medication if it contains valsartan unless they are told to stop by a doctor or pharmacist.
People are advised to speak to a pharmacist and find out if their medication is being recalled. If their medication is being recalled they should contact their doctor to discuss treatment options.
The recall impacts a number of countries including Germany, Norway, Finland, Sweden, Hungary, the Netherlands, Austria, Ireland, Bulgaria, Italy, Spain, Portugal, Belgium, France, Poland, Croatia, Lithuania, Greece, Canada, Bosnia and Herzegovina, Bahrain and Malta.
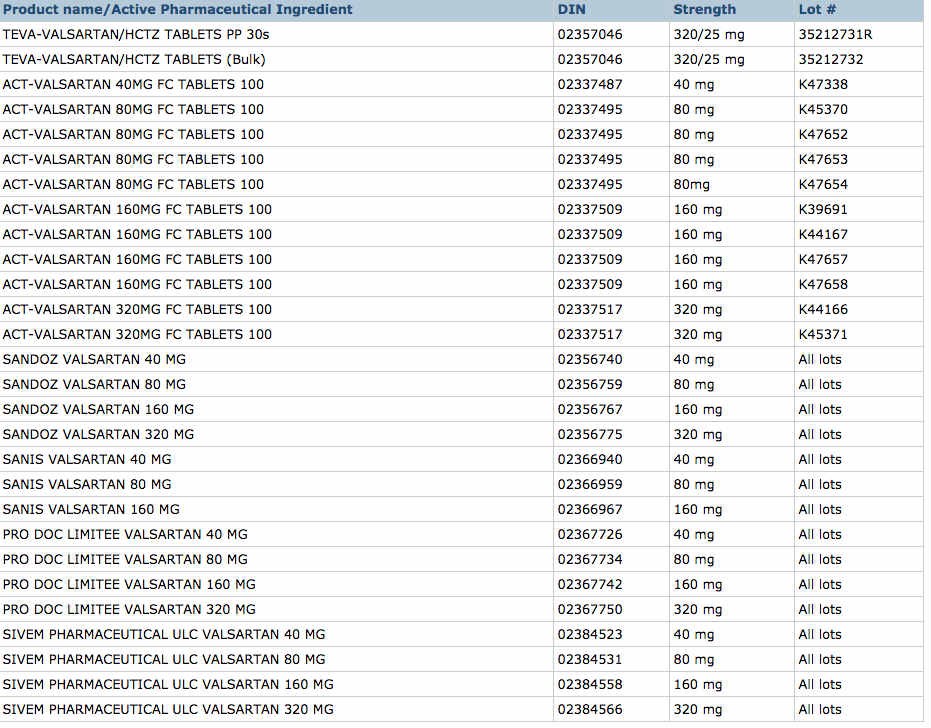
Zhejiang Huahai Pharmaceuticals in Linhai, China is the supplier of the contaminated valsartan and five companies have affected products that are being recalled, which are listed in the table below:
 Health Canada
Health Canada


